New smelting reduction process recovers Co, Ni, Mn, and Li from Li-ion batteries
A team from metals research institute SWERIM in Sweden has reported on a smelting reduction process to recover cobalt, nickel, manganese and lithium simultaneously from spent Li-ion batteries. A paper on their work was published in the Journal of Power Sources.
Results from the laboratory-scale smelting reduction (carried out at 1,550°
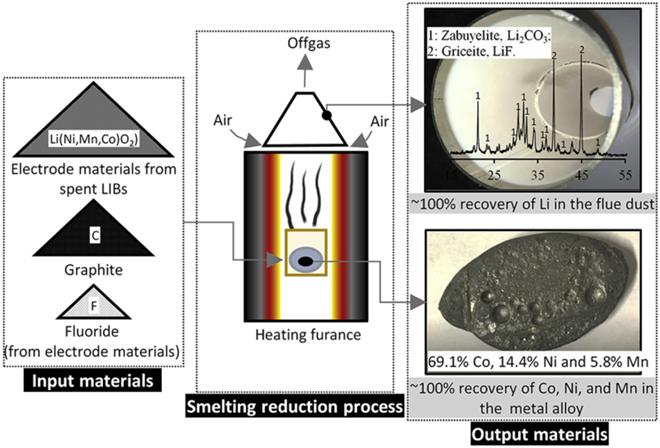
C in an Ar atmosphere in a vertical furnace) of chemical-grade LiCoO2 with and without the presence of halides (CaF2 and CaCl2) indicated that component Co2O3 in LiCoO2 could be reduced to Co metal. Meanwhile, component Li2O in LiCoO2 could be reduced to lithium metal vapor or converted into volatile lithium halides (LiF and LiCl) and subsequently be recovered in the flue dust.
The researchers say the results from smelting reduction of electrode materials of spent batteries indicate that the electrode materials could be smelted into Co-Ni-Mn alloys, while lithium can be concentrated and recovered in the flue dust as Li2CO3 and LiF. The absence of a slag allows a nearly 100% recovery of Co, Ni, and Mn in the formed alloy and a nearly 100% recovery of lithium in the flue dust.
Principal Investigator Xianfeng Hu summarized the team’s findings: “This paper provides technical information for developing a pyrometallurgical dominant process to recover Co, Ni, Mn, and Li from electrode materials of LIBs. Yet, the scales of the carried-out trials are rather small, and the obtained results are limited to the laboratory findings. Further, no slag-forming materials are added during the smelting reduction process. This is substantially different from an industrial smelting reduction process, in which a slag phase is present to prevent the oxidation of molten alloy and for refining purposes. To provide a better reference for the industrial implementation, the proposed smelting process needs to be demonstrated on a pilot scale. The results from a pilot-scale demonstration in an electric arc furnace are presented in a consecutive paper (Part II).”
Source: SWERIM via Green Car Congress
